A new view on the origin of cancer is proposed by Antonio Mazzocca and collaborators at the University of Bari "Aldo Moro" in a recently published paper in the journal "Medical Hypothesis"
The uncoupling of two subsystems evolutionarily conserved in eukaryotic cell pushes neoplastic transformation
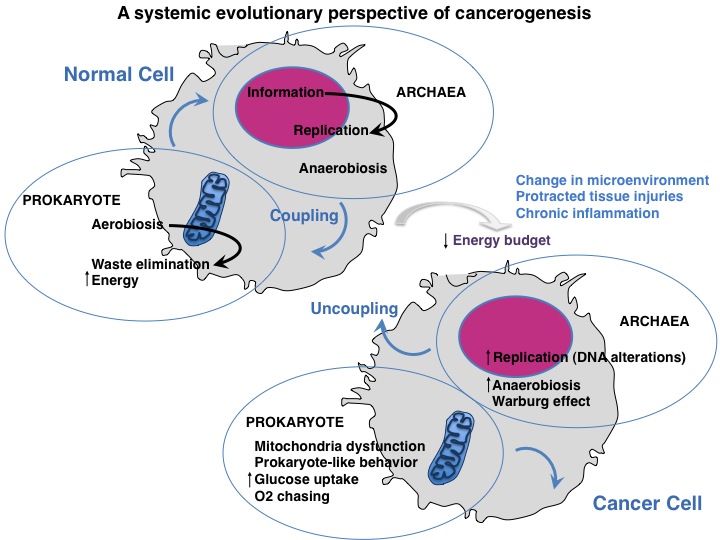
The authors of this study propose a systemic-evolutionary approach according to which, one of the thrust engines to neoplastic transformation would be the uncoupling of two "subsystems", evolutionarily conserved in eukaryotic cell: the informational part in the nucleus-cytoplasm (the ancestralarchaea) and the energy/mitochondrial part (the ancestral prokaryote). The integration of the two subsystems is ensured by the constant energy intake in normal condition which is narrowing due to organ damage
According to the theory proposed, these two components or "subsystems" are perfectly integrated into the cell and would require a determined and constant amount of energy in order to function properly under normal conditions. The differentiated state of the cell under normal conditions would thus be guaranteed by the constant energy intake in the form of "energy packs". By contrast, as a result of a prolonged insult or organ damage (i.e. chronic inflammation or fibrosis), which involves a reduced supply of oxygen and nutrients and therefore the reduction of energy at the cellular level, the two subsystems would face,over time, to a gradual decoupling process. In other words, when enduring insults compromise the cellular or tissue organization, the amount of energy needed to maintain the cell differentiation process can be limited and this may cause, over time, a state of de-differentiation or preneoplastic cell.
The multistep process of hepatocarcinogenesis as a paradigm
In the cirrhotic liver, which is an excellent paradigm, this process can be facilitated by the reduced availability of oxygen and nutrients to the organ due to an altered vascular system or by a "barrier" caused by fibrosis. In the long run, this would lead to a complete decoupling of the two subsystems, with the informational part (which becomes dysfunctional; DNA damage, etc.) dropped from the mitochondrial component (energy metabolism dysfunction), which lead to cell transformation. An example is liver cancer that arises in a cirrhotic liver. In fact, the entire process of hepatic carcinogenesis provides a study model that supports this hypothesis. When cancer occurs, the two "pre-eukaryotic subsystems" re-emerge becoming predominant, as shown by the metabolic abnormalities of cancer cells (predominantly anaerobic glycolysis and use of glutamine), and by their ability to proliferate and invade, behavior very similar to the two ancestral symbiotic components of the eukaryotic cell (the archaea and the prokaryotic). It is as if in the cancer cell prevailed a type of energy metabolism "microbial-like" or "prokaryotic-like ".
De-emergence of the eukaryotic system, and re-emergence of the two ancestrally preserved subsystems
This theory posits, in fact, the de-emergence of the "eukaryotic cell" system and the re-emergence of the archaea and prokaryotic subsystems, with an uncoordinated behavior. The decrease in the coordination of the two subsystems would be caused by alterations and impairments in the organization of the microenvironment tissue (mainly chronic inflammation, fibrosis, etc.), damage to nuclear and/or mitochondrial DNA (by viruses, chemicals, several agents including foodborne agents) which control the metabolic pathways or the mitochondrial energy production, etc., all risk factors known to favor the onset of cancer.
Unifying Theory and New Scenarios
This new theory could bridge the gap or unify the two theories about the origin of cancer prevalent today; theone referring to thesomatic mutations (SMT) and the otherone regarding the tissue organization field theory (TOFT) that identifies the basic defect in the cell-cell communication in a society of cells. In addition to opening a new avenue to the interpretation of the complexity of cancer, this new perspective marks a paradigm shiftthat could open scenarios for new therapeutic approaches.
Source: (http://www.sciencedirect.com/science/article/pii/S0306987716301578).
Antonio Mazzocca, M.D., Ph.D.
Interdisciplinary Departement of Medicine
University of Bari School of Medicine
Email: [email protected]
Website: http://www.antoniomazzocca.com/