Gene-edited (GEd) plants are developed using the advanced molecular biology techniques that may facilitate precise, efficient, and targeted modifications at genomic loci.The gene editing tools allow mutation, insertion, deletion, or substitution of DNA at targeted sites. Most common tool being employed in the development of GEd plants/ organisms includes clustered regularly interspaced short palindromic repeats (CRISPR)–CRISPR-associated protein (Cas9). Besides CRISPER-Cas9, transcription activator-like effector nucleases (TALENs), zinc-finger nucleases (ZFNs), and meganucleases have also been used (Zhang et al., 2018).
CRISPR-Cas9 system as a gene editing tool
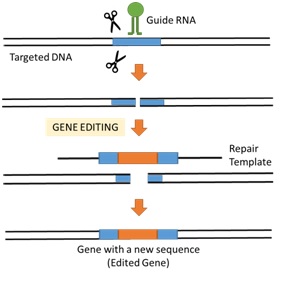
The CRISPR-Cas9 system uses site-directed nucleases to target and modify DNA with accuracy (Fig. 1). CRISPR is an integral part of a bacterial defense system, which forms the basis of the CRISPR-Cas9 system.
The gene editing through CRISPER-Cas9 can be achieved by following mechanisms:
- Gene knock-out: A single guide RNA (sgRNA) is used to target genes, which initiate a double stranded break using the Cas9 endonuclease. These breaks are then repaired by an innate DNA repair mechanism, resulting in genomic deletions or insertions,
- DNA-free gene editing: CRISPR can also be used for gene editing without the use of DNA vectors, requiring only RNA or protein components.
- Gene insertions or “knock-ins”: The CRISPR-induced double-strand break can also be used to create a gene “knock-ins” by using the cells’ homology-directed repair.
- Transient gene silencing:Transient gene silencing or transcriptional repression can be done by modifying the Cas9 protein,
|
Mechanism of Gene Editing of CRISPR/Cas9 |
Grouping of GEd plants
|
GEd Group I (SDN-1) |
GEd Group II (SDN-2) |
GEd Group III (SDN-3) |
|
Single or few base pair edits/deletions/insertions or small deletions (SDN-1) leading to least complexity (Phenotype/ Genotype).
Changes leading to knockdown/knock out of protein/ RNA that result in a new trait, which may befamiliar with prior knowledge. |
Several base pair edits (SDN-2) leading to certain degree of complexity in Phenotype/ Genotype (leading to improvement of an existing trait or creation of a new trait).
Changes leading to gain of function with a new protein or RNA that may or may not be familiar with prior knowledge. |
Insertion of foreign gene/DNA sequence (like SDN-3) leading to high degree of complexity in Phenotype/ Genotype (leading to creation of a new trait, new metabolic pathways, etc.).
Changes leading to gain of function with new protein or RNA that may not have prior knowledge.
|
Gene Editing vs Gene Modification
|
|
Gene Editing |
Gene Modification |
|
Introduction |
The gene editing tools allow mutation, insertion, deletion, or substitution of DNA at targeted sites for development of GEd plants. |
Genetically Modified (GM) Plants/ Organisms (GMOs) are developed by insertion of foreign gene cassette of desired trait employing recombinant DNA (rDNA) technology. |
|
Level of Modification |
The GEdplants, may contain very specific modification of one or few base pairs within the existing genetic information of living organisms with known genome structure and function without involving foreign gene insertion. |
Insertion of foreign gene in GMOs may lead to expression of new or recombinant proteins. |
|
Regulation |
Group I GEdplants are similar to naturally occurring variants in the cultivated germplasm, wild species and mutants generated through chemical/ radiation mutagenesis. In case of Group III GEd, there may be formation of new proteins due to foreign gene insertion, |
GMOs are regulated in most countries by biosafety frameworks established by specific legislation. In some countriessuch as in the United States, labelling of GM products isvoluntary; whereas it is mandatory in several countries asin the European Union (EU). |
References
- Zhang, Y., Massel, K., Godwin, I.D. et al. (2018) Applications and potential of genome editing in crop improvement. Genome Biol 19, 210. https://doi.org/10.1186/s13059-018-1586-y
- https://www.isaaa.org/resources/publications/pocketk/54/default.asp